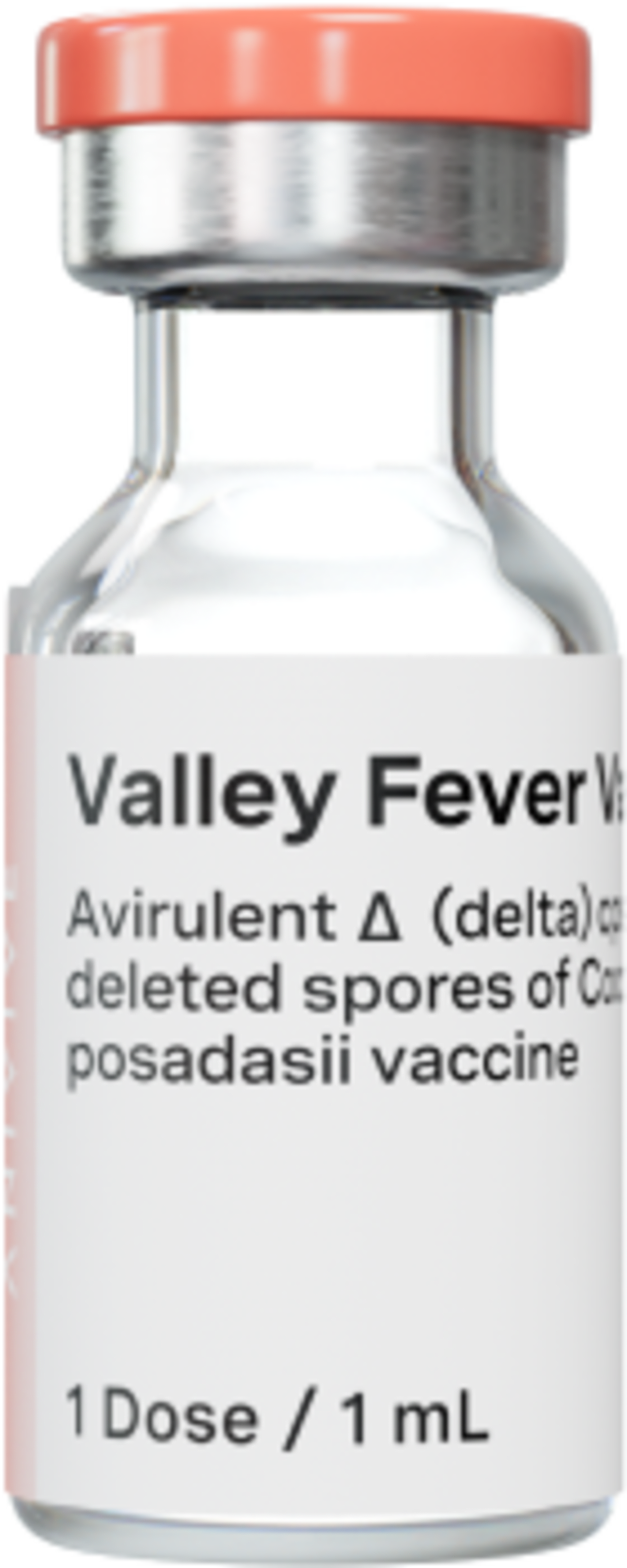
VALLEY FEVER VACCINE
Valley Fever is currently unpreventable - and spreading
Fighting Valley Fever
4-Time Super Bowl Champion and 5-Time NFL Pro-Bowler Rob Gronkowski, who played college football at the University of Arizona, has seen the impact of Valley Fever in the region. As the proud pet parent to his French Bulldog Ralphie, Gronk is advocating for pets and their owners to have access to affordable and innovative care.


Intelligent Software.
Better Treatments.

 Gather
Gather
The smartest research engine in pet health is populated with decades of global research and over 1 billion datapoints.
 Analyze
Analyze
Daily data and DNA contributions fuel the increasing intelligence of our AI-powered search for new ways to treat existing diseases.
 Develop
Develop
Decades of data on pet health and predictive analytics help us focus on the most promising new drugs for pets.
Industry leading technology and research to keep your pets healthy.
Targeted Medicine
For Unmet Needs
Targeted Medicine
For Unmet Needs
The discovery module identifies promising therapeutics for pets, then predicts how effective they'll be. The power of AI puts drug development on the fast track - efficiency that makes our medicines more affordable.
AI Imaging
For Fungal Diagnosis
The imaging module has ingested thousands of x-rays and can now detect systemic fungal infections in dogs. Read our research Detecting Valley fever with Deep Convolutional Neural Networks.
DNA Insights
With Carbon DNA Kit
The gene module flags key insights from your pet's DNA data. If your pet is prone to specific health issues, we'll point you to studies and actionable advice to help you stay ahead.
Connecting Pets with life saving medicine
 Marketplace
Marketplace


OUR PORTFOLIO
Treatments. Vaccines.
Novel Applications.
We're working on a variety of products all designed to address pet health specifically and expand treatment options currently available. The therapeutics in our pipeline today focus on cancer care, fungal diseases, viral infections, and more.
Valley Fever
CPS1USDA
FIP
INAD I-013287
Blastomycosis
BAD1USDA
Hemangiosarcoma
INAD I-013225
Gingivitis
Lysine Decarboxylase
Osteosarcoma
INAD I-013511Products in development not approved by the FDA or USDA
OUR SERVICES
Bespoke tools for
Veterinary Professionals
We're transforming pet pharma forever.


Man versus mycelium: The race to create vaccines against fungal threats

How research of an FIP treatment for cats is saving human lives


Petco Love Joins Forces With Anivive Lifesciences and Rob Gronkowski to Champion The Fight Against Valley Fever
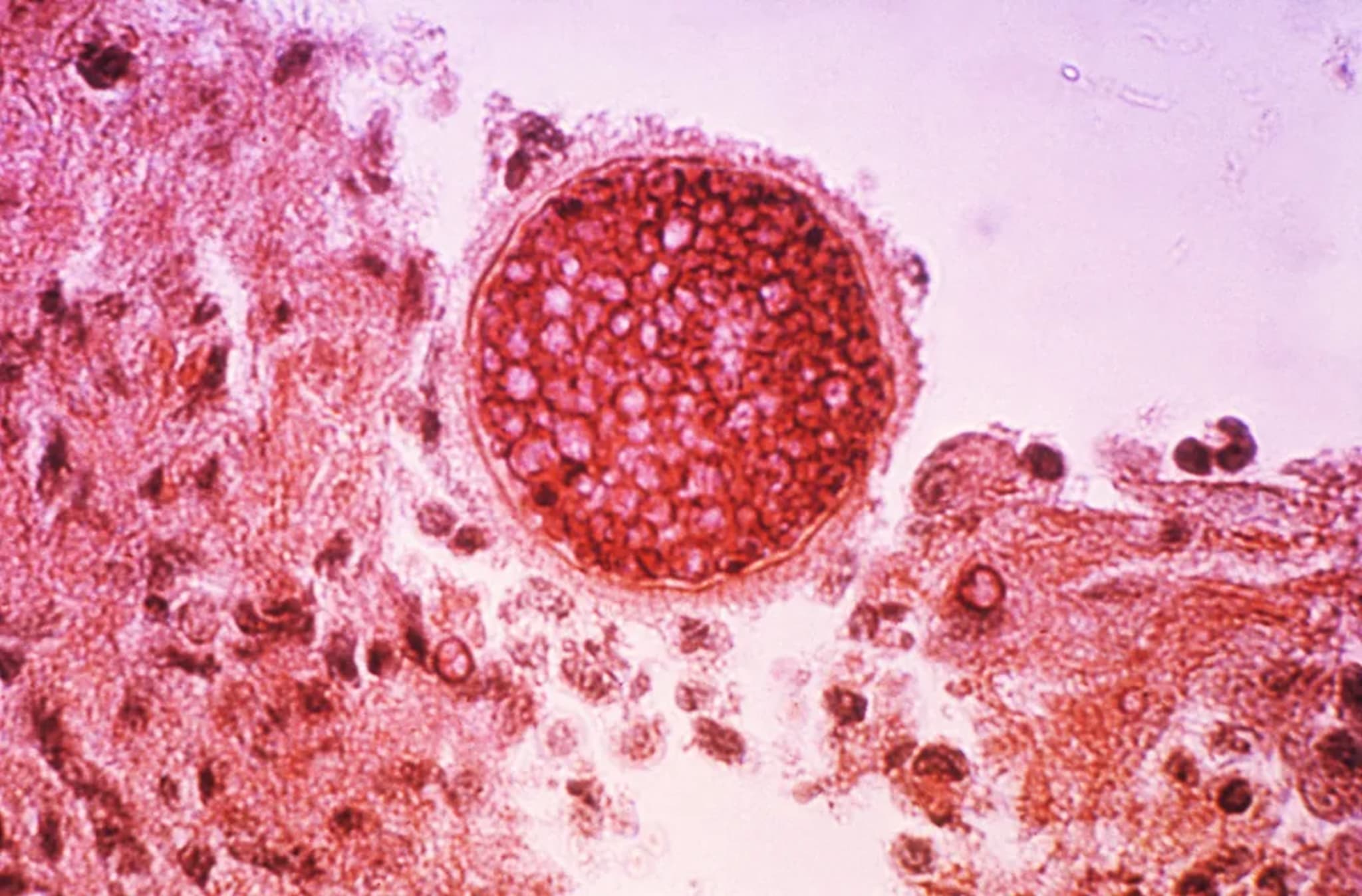

A Vaccine Against Valley Fever Finally Works—for Dogs


Addressing therapeutic needs in veterinary health


Getting closer to a vaccine to prevent Valley Fever in dogs
The team revolutionizing
pet health

Scott Whitcup

James Beaubien

Dylan Balsz

David Bruyette DVM

Glenn Stearns

Lynne Doti PhD

Robert Bishop PhD

Scott Whitcup

James Beaubien